In Silico Vagus Nerve Stimulator: Design, Optimization, and Safety Evidence Generation
by IT’IS FoundationMulti-Scale Geometric Modeling
The vagus nerve model was derived from segmented cross-sectional histology data and included:- multiple fascicles,
- epineurium, perineurium, and endoneurium layers,
- conductive, microstructured electrode contacts on a micrometers-thin polyimide substrate,
- the surrounding environment (differs between in vitro and in vivo setup).
The geometry spans millimeter-scale fascicular structures and micrometer-scale electrode contacts and insulation layers, requiring careful multi-scale discretization. The 2D segmentation was extended to a 3D model using controlled extrusion and Sim4Life's unstructured multi-domain meshing, with explicit refinement in thin layers and near electrode contacts.

Discretization of nerve cross-section and 2D mesh refinement for convergence analysis.
Electromagnetic Simulation and Convergence
Electro-quasistatic simulations were performed using low-frequency conductivities from the curated, quality-assured IT'IS Tissue Properties Database . Mesh densities ranging from approximately 30 to 120 million cells were evaluated. Convergence analysis focused on:- electric field magnitude,
- electric field gradients along representative axonal trajectories,
- stability of predicted activation thresholds under mesh refinement.
This ensured that recruitment predictions were not artifacts of discretization.
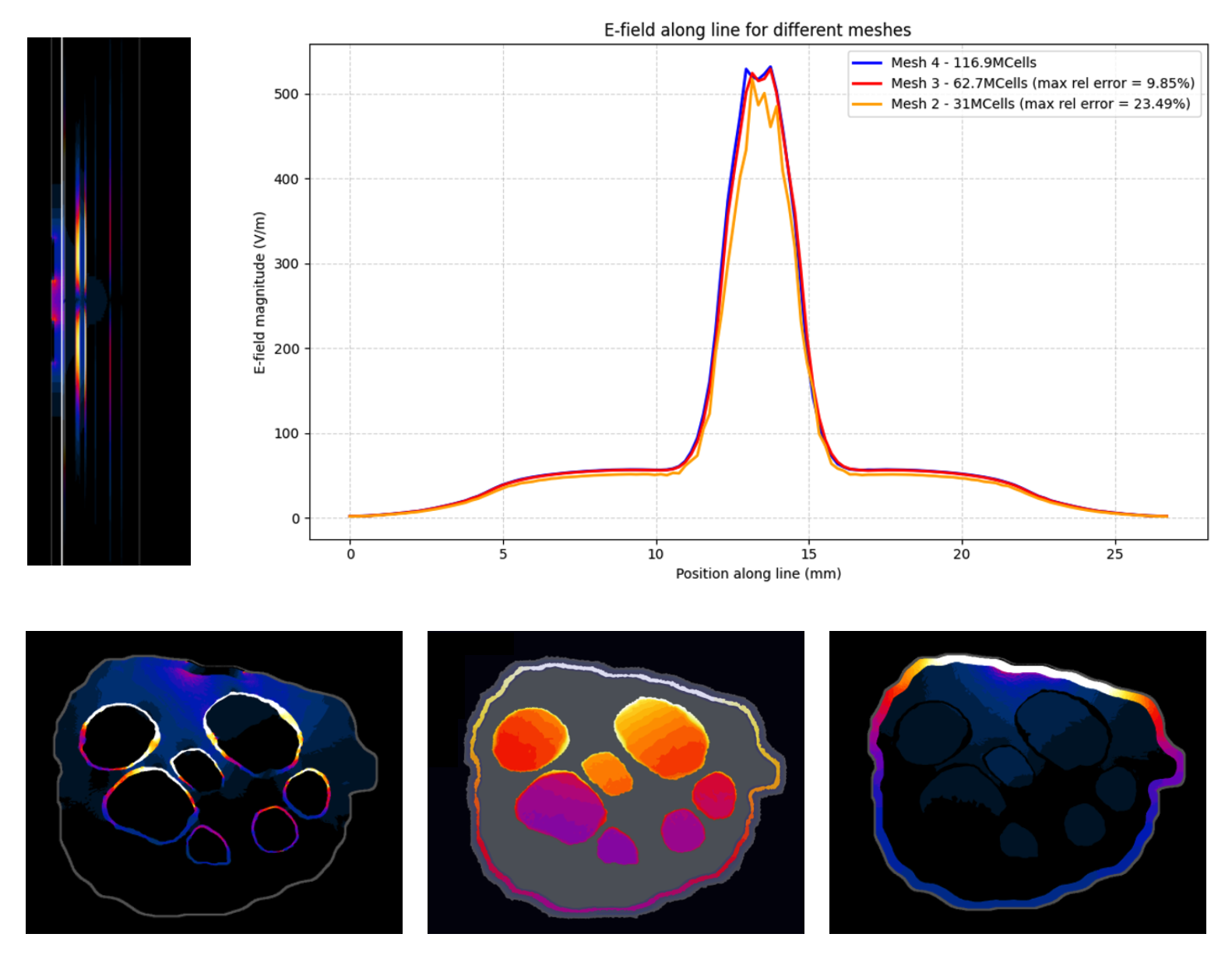
Electromagnetic simulation results and mesh convergence analysis. Top right: Electric field interpolated along a line within the nerve for the three meshes with different refinement levels, enabling comparison of the results and assessment of mesh convergence. Left: Electric field distribution along the longitudinal section of the nerve, showing higher field intensities in regions closer to the electrodes. Bottom: Cross-sectional views showing (a) the electric field distribution, with higher field values in the perineurium in regions closer to the electrodes (top fascicles); (b) the electric field gradient, which is the quantity most relevant for axonal activation, here stronger in regions closer to the electrodes (top fascicles); and (c) the current density distribution, confirming that current remains confined within the nerve and does not flow outward.
Neurofunctionalization and Recruitment Analysis
Fascicles were populated with predefined, parameterized Sim4Life models of myelinated and unmyelinated axons, using diameter distributions derived from human biopsy data. Recruitment curves were computed to determine stimulation currents corresponding to defined recruitment and conduction block levels. The results demonstrated a nonlinear relationship between applied current and fiber activation, reflecting the spatial heterogeneity of field gradients within the nerve.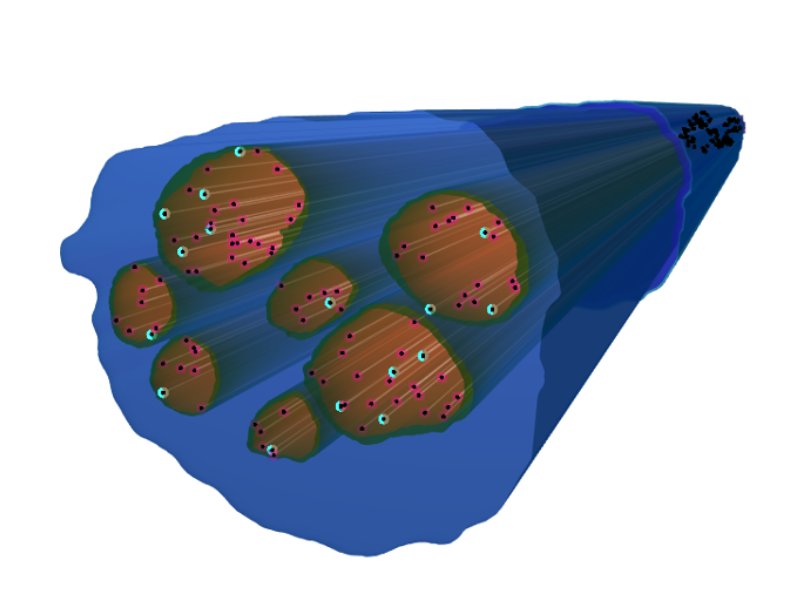
The blue volume represents the 2.5D model of the nerve, containing multiple fascicles shown in brown. Within each fascicle, colored points indicate the spatial distribution of simulated axons: pink points represent unmyelinated axons, while cyan points represent myelinated axons. The figure illustrates an example population of 100 axons distributed within the fascicles, composed of 20% unmyelinated and 80% myelinated fibers.
Thermal Modeling and Safety Assessment
Temperature rise from electric power deposition was assessed using Sim4Life's Bioheat equation simulator. As with the electromagnetic simulations, a careful convergence analysis was performed, along with an investigation of domain truncation effects. Sim4Life's thermal dose modeling was then used to translate transient temperature evolution into metrics comparable to tissue damage thresholds, ensuring sufficient safety margins.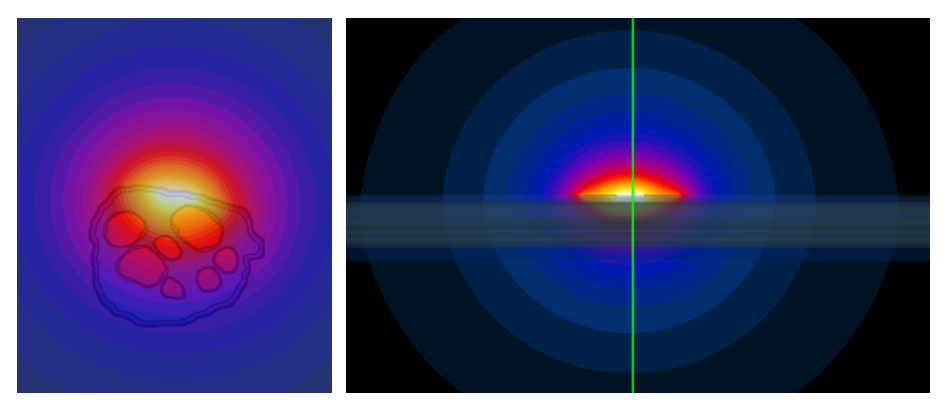
Thermal simulation results based on electromagnetic field distribution. Temperature rise distribution obtained from the thermal simulation using the electromagnetic field as the heat source. The left panel shows cross-sectional views of the nerve, while the right panel shows the longitudinal section. The results illustrate that the highest temperature increase occurs in the region surrounding the electrodes and gradually propagates into the nerve and the surrounding tissue (muscle in this model). The temperature distribution decreases radially with distance from the stimulation site.
Parameterized Modeling, Design Optimization, and Uncertainty Quantification
The entire modeling pipeline was converted into a parameterized study, enabling the application of Sim4Life's Model Intelligence Hypertools . Surrogate-modeling-based multi-objective optimization was performed, weighing 11 safety and effectiveness metrics to identify superior electrode designs – maximizing effectiveness and efficiency while minimizing risk metrics such as tissue heating and the Shannon criterion for electrochemical damage.Adaptive surrogate modeling was then used for comprehensive uncertainty quantification, propagating tissue property and numerical uncertainties through the nonlinear model.